
Get Compensated
for Your Time

Participating in clinical trials takes time and Vial makes sure that you are compensated for that time.
Сlinic not defined
Please check back at a later date, or fill out a one-minute form to have your information added to a waiting list.
GET ON VIAL'S WAITLISTThis form helps us determine if there is a trial that you could be eligible for.
During a brief phone call, we will give you more details about the study, discuss your medical history, and book your appointment at the clinic if you qualify.
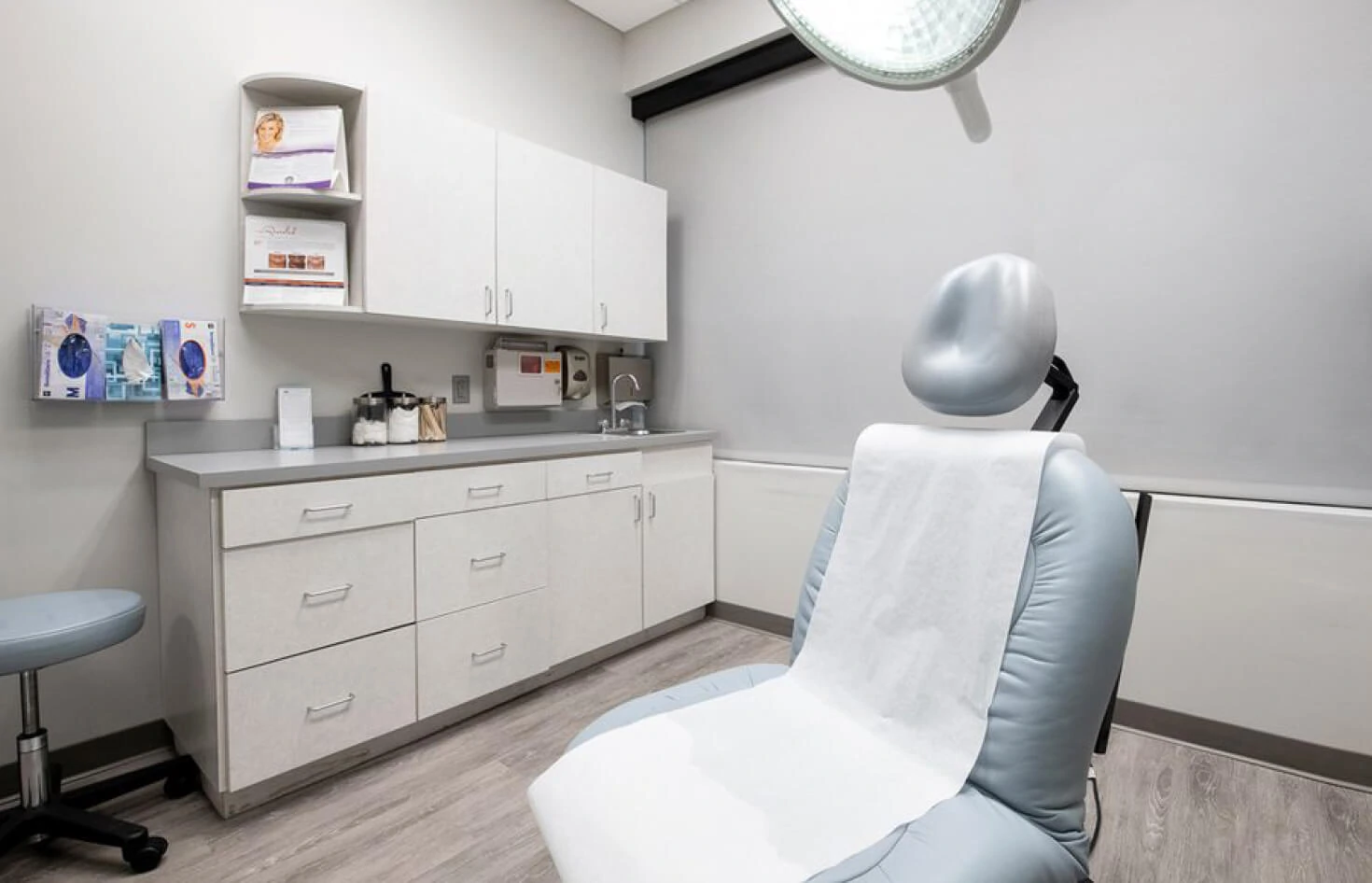
Visit our clinic for your screening visit. You will be seen by the doctor as well as the clinical research coordinator.
Meet Your Doctor


Participating in clinical trials takes time and Vial makes sure that you are compensated for that time.
Between doctor visits, prescriptions, and trips to the drug store, it may be tough to find an effective treatment for your specific skin condition. When you participate in a Vial trial, you may get access to new investigative treatments at no cost.



Pick a time and date that works for you to come visit one of our conveniently located clinics.
If you do not find an answer to your question, please call us
Сlinic not defined
If you do not find an answer to your
question, please call us Сlinic not defined
Before signing up for a study, someone will review the informed consent form with you. This important step makes sure you have all the information you need to decide whether to join. After you sign the form and join the study, you will begin receiving treatment related to the study. This may seem like getting normal treatment, but you may be asked additional questions or expected to visit the clinic more often than you would for normal treatment. You may also receive different medication than you’re used to or may receive a placebo product. Communication between you and the study team is important, so you may also receive updates and reminders about your treatment or upcoming clinic visits. At all times your health and safety will be closely monitored.
The medication you might receive will vary depending on the study that you participate in. The medication is developed by pharmaceutical companies who are sponsoring the clinical trial and it’s important to understand the medication is still an investigational product. During the enrollment process, someone will give you more information about the trial, including the medication that you might receive and any related risks. You will have an opportunity to ask questions and make sure you’re comfortable before proceeding with participation in the study.
Depending on which study you join, you may be eligible for a patient stipend payment which compensates you for your time and effort related to participation. You will also receive the study medication and related care at no cost to you.
You do not have to pay for any treatment related to the clinical trial including the study medication. In fact, some studies may also offer patient stipends that compensate you for your time and effort related to participation in the study.
When you submit an enrollment request on our website, we will ask you some questions in the sign up form to see if you meet basic requirements for the particular study. If you do, someone will give you a call to ask more detailed questions that can be answered remotely. However, there are some eligibility requirements that can only be determined with a visit to the clinic. The questions we ask are important to make sure you can safely participate in the trial. After your first visit to the clinic, you will know if you are fully eligible for the trial.
The number of visits to the clinic varies depending on which study you are participating in, but generally ranges from 2-12 visits. Prior to you enrolling in the study, someone will explain how many visits are required so you can decide whether to join.
Yes. All studies are completely voluntary and if at any time before the conclusion of the study you want to withdraw your participation, you are completely free to do so.
Please let us know if you require assistance getting to the clinic; we may be able to help.
You will be seen by a doctor at the clinic. There might also be some other staff members at the clinic there to help out with the trial. Everyone at the clinic is trained to be professional and treat you with the utmost respect.
Any information you share with Vial is handled by us according to our Privacy Policy. When you join a study, you will also be provided with detailed information about how your information in the study is used.