Sevoflurane
Back to searchMolecule Structure
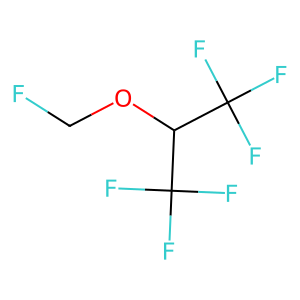
Scientific Name
Sevoflurane
Description of the Drug
Sevoflurane is a inhalation anaesthetic agent used for induction and maintenance of general anesthesia during surgical procedures.
Source: DrugBank Online – DrugBank.com. Retrieved 2023-03-10 from
http://www.drugbank.ca/drugs/DB01236
http://www.drugbank.ca/drugs/DB01236
Brand Name(s)
Sevoflurane, Sojourn, Ultane
Company Owner(s)
Piramal Critical Care Inc, Abbvie Inc, Halocarbon Products Corp, Shanghai Hengrui Pharmaceutical Co Ltd, Baxter Healthcare Corp
Mechanism(s) Of Action
| Target Name | Target Type | Action Type | Target ChEMBL ID |
|---|---|---|---|
| Potassium channel subfamily K member 9 | SINGLE PROTEIN | OPENER | CHEMBL2321614 |
| Potassium channel subfamily K member 2 | SINGLE PROTEIN | OPENER | CHEMBL2321615 |
| Potassium channel subfamily K member 3 | SINGLE PROTEIN | OPENER | CHEMBL2321613 |
| Potassium channel subfamily K member 10 | SINGLE PROTEIN | OPENER | CHEMBL2331041 |
| Glycine receptor (alpha-1/beta) | PROTEIN COMPLEX | POSITIVE MODULATOR | CHEMBL2363052 |
| Potassium channel subfamily K member 18 | SINGLE PROTEIN | OPENER | CHEMBL2331042 |
| GABA-A receptor; anion channel | PROTEIN COMPLEX GROUP | POSITIVE ALLOSTERIC MODULATOR | CHEMBL2093872 |
Clinical Trial(s)
Unichem Links
| Atlas | sevoflurane | baseline sevoflurane | ||
| SureChEMBL | SCHEMBL61918 | |||
| PharmGKB | PA451341 | |||
| Human Metabolome Database | HMDB0015366 | |||
| DrugBank | DB01236 | |||
| PubChem: Thomson Pharma | 15120951 | |||
| PubChem | 5206 | |||
| Nikkaji | J20.100F | |||
| EPA CompTox Dashboard | DTXSID8046614 | |||
| DrugCentral | 2439 | |||
| ChemicalBook | CB1440979 | |||
| Guide to Pharmacology | 7296 | |||
| rxnorm | SEVOFLO | SEVOFLURANE | ULTANE | SOJOURN |
| PubChem: Drugs of the Future | 12012762 | |||
| KEGG Ligand | C07520 | |||
| ChEBI | 9130 | |||
| ZINC | ZINC000001530810 |