Glyburide
Back to searchMolecule Structure
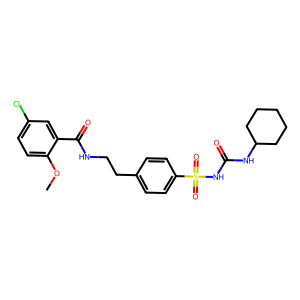
Scientific Name
Glyburide
Description of the Drug
Glyburide is a sulfonylurea used in the treatment of non insulin dependent diabetes mellitus.
Source: DrugBank Online – DrugBank.com. Retrieved 2023-03-10 from
http://www.drugbank.ca/drugs/DB01016
http://www.drugbank.ca/drugs/DB01016
Brand Name(s)
Calabren, Daonil, Diabeta, Diabetamide 2.5, Diabetamide 5, Euglucon, Gliken, Glyburide, Glyburide (micronized), Glynase, Glynase Prestab, Lederglib, Libanil, Malix, Micronase, Semi-daonil
Company Owner(s)
Epic Pharma Llc, Teva Pharmaceuticals Usa Inc, Impax Laboratories Inc, Bristol Myers Squibb, Chartwell Rx Sciences Llc, Orient Pharma Co Ltd, Actavis Elizabeth Llc, Heritage Pharmaceuticals Inc Dba Avet Pharmaceuticals Inc, Hikma Pharmaceuticals, Aurobindo Pharma Ltd
Mechanism(s) Of Action
| Target Name | Target Type | Action Type | Target ChEMBL ID |
|---|---|---|---|
| Sulfonylurea receptor 1, Kir6.2 | PROTEIN COMPLEX | BLOCKER | CHEMBL2096972 |
Clinical Trial(s)
Unichem Links
| Atlas | glibenclamide | ||
| SureChEMBL | SCHEMBL22009 | ||
| PharmGKB | PA449782 | ||
| Human Metabolome Database | HMDB0015151 | ||
| DrugBank | DB01016 | ||
| PubChem: Thomson Pharma | 14835352 | ||
| PubChem | 3488 | ||
| Mcule | MCULE-2351642942 | ||
| LINCS | LSM-2811 | ||
| Nikkaji | J3.430D | ||
| PDBe | GBM | ||
| BindingDB | 50012957 | ||
| EPA CompTox Dashboard | DTXSID0037237 | ||
| DrugCentral | 1314 | ||
| Brenda | 232858 | 14374 | 206663 |
| ChemicalBook | CB1737679 | ||
| Guide to Pharmacology | 2414 | ||
| rxnorm | GLYNASE | GLYBURIDE | |
| KEGG Ligand | C07022 | ||
| ChEBI | 5441 | ||
| ZINC | ZINC000000537805 |